Non-aqueous phase organic compounds
Some organic compounds have such a low solubility in water that they can be found below the ground in the non-aqueous phase, i.e. not dissolved in water.
The behaviour of low solubility organic compounds in the subsurface environment depends on whether they have a lower or higher density than water.

Light non-aqueous phase liquids (LNAPL)
Light non-aqueous phase liquids (LNAPL) have a lower density than water. Once LNAPL infiltrate to the subsurface, they are commonly retained close to the water table, within the zone of fluctuation.
The effort to locate and remove LNAPL is relatively cheaper and easier than DNAPL.
Examples of LNAPLs are petrol, diesel and other hydrocarbons.
Dense non-aqueous phase liquids (DNAPL)
Dense non-aqueous phase liquids (DNAPL) are denser than water and tend to sink below the water table when spilled in significant quantities.
Their penetration into an aquifer makes them difficult to locate and remediate.
DNAPLs come to rest in the subsurface as disconnected blobs and ganglia of liquid, and in potentially mobile distributions referred to as pools.
Once DNAPL is present in bedrock, it will slowly dissolve into groundwater, giving rise to aqueous phase plumes. The plumes will generally migrate in the hydraulically down-gradient direction subject to advection, dispersion, sorption to fracture walls, possible biodegradation and matrix diffusion.
Examples of materials that are DNAPLs when spilled include: chlorinated solvents, such as trichloroethylene, tetrachloroethene, 1,1,1-trichloroethane and carbon tetrachloride, coal tar, creosote and polychlorinated biphenyl (PCBs).
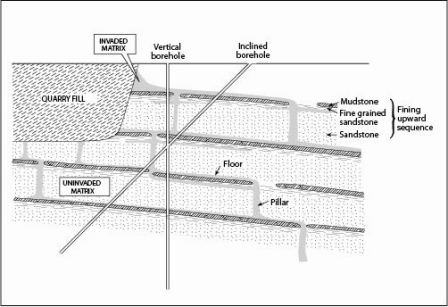
Research into chlorinated solvents
A study of chlorinated solvent distribution in the Permo-Triassic sandstone of north-west England showed that significant changes in lithology and permeability on the sub-metre scale control the distribution of solvents in the unsaturated zone (Lawrence et al., 2006).
These compounds may also be volatile and give rise to vapour plumes in the unsaturated zone.
Although chlorinated solvents tend to be relatively resistant to degradation within most aquifer systems, there can be effective degradation in anaerobic systems and under suitable conditions, biodegradation and volatilisation can contribute significantly to the removal of chlorinated solvents from the subsurface, making natural attenuation a potentially important remediation alternative.
The BGS was part of an international, multidisciplinary team which investigated these issues in the Source Area in situ BioREmediation (SABRE) project.
Contact
Contact Dan Lapworth for further information