Isotopic fingerprint of phosphorus in drinking water

We have, for the first time, characterised the isotopic composition of phosphorus in drinking water. Seventy per cent of samples were found to have a composition that could be distinguished from other sources of phosphorus in the environment. This research could lead to improved understanding of the relative importance of phosphorus sources in our freshwater and marine environment, in particular determining whether leakage from the drinking water supply network is a significant source of phosphorus. This could assist attempts to halt eutrophication of water bodies.
Protective phosphorus in drinking water
Phosphorus is naturally present in most waters. It is also added to 95 per cent of UK drinking waters in order to reduce the amount of lead that can dissolve into our water. This dosing of the water leads to concentrations of about 1 mg/l phosphorus in UK drinking water.
Lead intake is associated with health problems. Since the removal of lead from petrol, old lead drinking water pipes have become a major source of lead. To reduce this threat, public water utilities in the UK, parts of Europe and North America routinely dose drinking water supplies with a form of phosphorus known as phosphate which forms a coating on the inside of the pipe. This prevents lead in pipes from dissolving and is a highly successful way to reduce lead levels in drinking water.
Phosphorus causing environmental harm
Phosphorus from human sources causes eutrophication of rivers, lakes and coastal waters. Phosphorus is a nutrient, and adding it to water can allow unusual quantities of algae to grow in the water. When the algae die, the process of decomposition removes oxygen from the water, which can lead to deaths of other water life including fish.
Efforts to reduce phosphorus inputs to the water environment to prevent eutrophication have tended to focus on reducing inputs from farming and water treatment works.

Leaking drinking water as a phosphorus source
About 25 per cent of drinking water is lost from the drinking water distribution system through leakage, which equates to about 40 litres per customer each day. Thus some 1200 tonnes of phosphorus is entering the environment via this route. What is currently unknown is how much of this is entering the water environment and contributing to eutrophication.
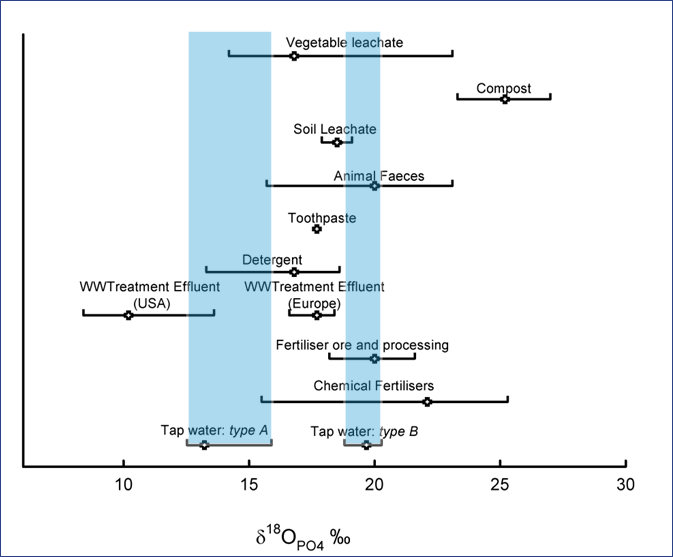
Stable oxygen isotopes of phosphate
We collected 40 samples of drinking water from water companies across England and Wales, and samples of the two sources of phosphorus compounds used to dose drinking water (types A and B). These were analysed via a 16 step process to determine the stable oxygen isotope ratio of the oxygen within phosphate molecules (δ18OPO4).
The oxygen isotope composition of phosphate from 70 per cent of the samples (type A) was found to be distinct from other sources of phosphorus to the environment (see Figure 1). The two sources of phosphate used by water companies have different isotopic compositions: type A is made from Chinese phosphatic rocks, while type B is from Morocco.
These findings can feed into future research to investigate what happens to the phosphorus from drinking water leakage. It may also be possible to identify phosphate in our rivers that has come from leaking mains, rather than from the other two major sources. Indeed there is also potential to differentiate between fertilisers and waste water. In the future this may mean we have to reconsider which environmental sources of phosphate require the greatest attention if we are to continue to reduce overall inputs to our rivers. The latest technical advancements give us some of the tools necessary to do this.
References
Gooddy, D C, Lapworth, D J, Ascott, M J, Bennett, S A, Heaton, T H E, and Surridge, B W J. :2015. Isotopic fingerprint for phosphorus in drinking water supplies. Environmental Science & Technology. DOI: 10.1021/acs.est.5b01137
Davies, C L, Surridge, B W J, and Gooddy, D C. 2014. Phosphate oxygen isotopes within aquatic ecosystems: global data synthesis and future research priorities. Science of the Total Environment, 496, 563–575. DOI: 10.1016/j.scitotenv.2014.07.057
Contact
Contact BGS Enquiries for further information.